Fri, May 8, 2026
[Archive]
Volume 11, Issue 2 (May 2026)
JNFS 2026, 11(2): 178-190 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Goodarzi S, Mirjalili F, Forootani B, Yekrang Safakar H, Salehi-Abargouei A. Polyphenol Intake and the Risk of Pancreatic Cancer: A Systematic Review and Meta-analysis of Observational Studies. JNFS 2026; 11 (2) :178-190
URL: http://jnfs.ssu.ac.ir/article-1-1291-en.html
URL: http://jnfs.ssu.ac.ir/article-1-1291-en.html
Sima Goodarzi 
 , Fatemeh Mirjalili
, Fatemeh Mirjalili 
 , Bita Forootani
, Bita Forootani 
 , Hooman Yekrang Safakar
, Hooman Yekrang Safakar 
 , Amin Salehi-Abargouei *
, Amin Salehi-Abargouei * 


 , Fatemeh Mirjalili
, Fatemeh Mirjalili 
 , Bita Forootani
, Bita Forootani 
 , Hooman Yekrang Safakar
, Hooman Yekrang Safakar 
 , Amin Salehi-Abargouei *
, Amin Salehi-Abargouei * 

Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran
Full-Text [PDF 649 kb]
(46 Downloads)
| Abstract (HTML) (802 Views)
1 Nutrition Research Center, Shiraz University of Medical Sciences, Shiraz, Iran; 2 School of Nutrition and Food Sciences، Shiraz University of Medical Sciences, Shiraz, Iran; 3 Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran; 4 Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran; 5 Students’ Research Committee, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
.PNG)
.PNG)
Full-Text: (6 Views)
Polyphenol Intake and the Risk of Pancreatic Cancer: A Systematic Review and Meta-analysis of Observational Studies
Sima Goodarzi; PhD Candidate3-5, Fatemeh Mirjalili; PhD Candidate3-5, Bita Forootani; MSc3,4, Hooman Yekrang Safakar; MSc3,4 & Amin Salehi-Abargouei; PhD*1-4
Sima Goodarzi; PhD Candidate3-5, Fatemeh Mirjalili; PhD Candidate3-5, Bita Forootani; MSc3,4, Hooman Yekrang Safakar; MSc3,4 & Amin Salehi-Abargouei; PhD*1-4
1 Nutrition Research Center, Shiraz University of Medical Sciences, Shiraz, Iran; 2 School of Nutrition and Food Sciences، Shiraz University of Medical Sciences, Shiraz, Iran; 3 Research Center for Food Hygiene and Safety, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran; 4 Department of Nutrition, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran; 5 Students’ Research Committee, School of Public Health, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
| ARTICLE INFO | ABSTRACT | |
| ORIGINAL ARTICLE | Background: Considering the antioxidant effects of polyphenols, it remains unclear whether dietary polyphenols can affect the risk of pancreatic cancer. This study assessed the association between polyphenol intake and the risk of pancreatic cancer. Methods: Articles published until April 2023 in PubMed, Scopus, and the Web of Science (ISI) were searched. Observational studies on the association between dietary polyphenol intake and the odds of pancreatic cancer were also included. Odds ratios (ORs) with 95% confidence intervals (CIs) were used as effect sizes. Furthermore, standard methods were used to evaluate the heterogeneity, sensitivity, and publication bias. The Newcastle-Ottawa Scale (NOS) was used for quality assessment. Results: Eight studies were included. Although this systematic review and meta-analysis revealed no relationship between the intake of total flavonoids, flavan-3-ols, flavones, flavonols, flavanones, anthocyanidins, and pancreatic cancer odds; in qualitative analysis, a positive relationship was observed between genistein and the aflavin intake and pancreatic cancer risk. Conclusions: No relationship was observed between polyphenol intake and pancreatic cancer odds. Polyphenols have poor bioavailability and bioaccessibility, which may have affected their results. Therefore, more high-quality studies with precise designs are required to determine whether a relationship exists. | |
| Article history: Received: 10 Mar 2025 Revised: 15 Jun 2025 Accepted: 25 Aug 2025 |
||
| *Corresponding author: abargouei@gmail.com Department of Nutrition, School of Public Health, Sahid Sadoughi University of Medical Sciences, Yazd, Iran. Postal code: 8915173160 Tel: +98 35 31492229 |
||
| Keywords Polyphenol; Flavonoid; Pancreatic cancer; Observational studies. |
Introduction
Pancreatic cancer is the 12th most common malignancy and the 7th leading cause of cancer-related mortality worldwide (Bray et al., 2018). In the last two decades, the incidence of pancreatic cancer has doubled each year from 1990 to 2017. There was an increase in the number of deaths for both sexes from 196,000 in 1990 to 441,000 in 2017 (Pourshams et al., 2019). This disease is highly aggressive and has a poor survival rate, which imposes a critical global burden (Maisonneuve and Lowenfels, 2010). Failure to treat pancreatic cancer is due to the lack of appropriate screening and diagnostic methods, the location of the pancreas, the difficulty of biopsy from the pancreatic tissue, the aggressive nature of this disease, and the low response to chemotherapy and radiotherapy (Maisonneuve and Lowenfels, 2010). Therefore, prevention is the best choice to reduce the incidence and mortality rates of pancreatic cancer. Modifiable risk factors, including smoking, alcohol consumption, obesity, and diet, are associated with increased disease risk (Klein, 2021, Rawla et al., 2019). It has been shown that dietary factors affect pancreatic cancer by more than 30%, and there is evidence that certain foods can increase the risk of pancreatic cancer, while others may protect against it (Maisonneuve and Lowenfels, 2015, Michaud et al., 2005, Midha et al., 2016). Several observational studies have shown an inverse association between fruits and vegetables and pancreatic cancer (Anderson et al., 2009, Chan et al., 2005, Ghadirian and Nkondjock, 2010, Jansen et al., 2011, Liu et al., 2014, Wu et al., 2016). Fruits and vegetables are rich in antioxidants and anti-inflammatory compounds. Therefore, their intake might reduce oxidative stress, inflammation, and cancer risk (Liu, 2013, Slavin and Lloyd, 2012). Polyphenols are a large family of phytochemicals found in a wide variety of fruits, vegetables, flowers, and leaves (David et al., 2016, Miean and Mohamed, 2001, Rawla et al., 2019, Slimestad et al., 2020, Tian and Liu, 2020). Polyphenols have been investigated for their beneficial effects on several diseases and their symptoms (Bao et al., 2020, Goñi Cambrodón and Hernández Galiot, 2019, Lichota et al., 2019, Sanches-Silva et al., 2020, Serra et al., 2020, Singh et al., 2020).
The results of studies on the effects of polyphenols on pancreatic cancer are contradictory (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Mouria et al., 2002, Vuong et al., 2014, Yamagiwa et al., 2020). In 2002, Mouria et al. reported that food-derived polyphenols inhibited the growth of pancreatic cancer cells and prevented metastasis by causing mitochondrial dysfunction, which led to the release of cytochrome c, caspase activation, and apoptosis (Mouria et al., 2002). Arem et al. studied 2,379 patients diagnosed with pancreatic cancer. This study failed to support the hypothesis of an association between the total intake of polyphenols or any type of flavonoid and the risk of pancreatic cancer (Arem et al., 2013). Furthermore, Molina-Molina-Montes et al. reported no association between the intake of flavonoids, flavonoid subclasses, or lignans and pancreatic cancer risk (Molina-Montes et al., 2016).
To the authors’ knowledge, no systematic review or meta-analysis has been conducted to assess the association between polyphenol intake and the risk of pancreatic cancer. Therefore, this study aimed to investigate the association between polyphenol intake and pancreatic cancer odds by conducting a systematic review and meta-analysis of observational studies.
Materials and Methods
Study protocol
This review was reported based on the Meta-Analysis of Observational Studies in Epidemiology (MOOSE) (Stroup et al., 2000) and Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines (Moher et al., 2010). The protocol was registered in PROSPERO (International Prospective Register of Systematic Reviews, registration number: CRD42023487302).
Search strategy
Articles published until April 2023 in PubMed, Scopus, and the Web of Science (ISI) were searched. The inclusion and inclution criteria were shown in Table 1. There were no restrictions on the language or publication dates. Furthermore, to find any other related articles, a manual, comprehensive search in Google Scholar and searches of the references of related published articles were performed. The search was updated to August 2024 to include any relevant published studies.
Eligibility criteria
Two independent researchers (Forootani B. and Yekrang Safakar H) screened the relevant studies according to the eligibility criteria, and contradictions were resolved by a third investigator (Salehi-Abargouei A). Studies were included in this review if they 1) were a prospective cohort (cohort, case-cohort, or nested case-control), case-control, or cross-sectional; 2) investigated the association between polyphenol intake as exposure and pancreatic cancer odds as an outcome; 3) were performed in adults; and 4) had enough information about the desired outcomes (odds ratio (OR), relative risk (RR), hazard ratio (HR), and 95% confidence interval (CI)) for polyphenols of interest. Moreover, animal studies, randomized control trials, and studies without the necessary data for extraction were excluded.
The results of studies on the effects of polyphenols on pancreatic cancer are contradictory (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Mouria et al., 2002, Vuong et al., 2014, Yamagiwa et al., 2020). In 2002, Mouria et al. reported that food-derived polyphenols inhibited the growth of pancreatic cancer cells and prevented metastasis by causing mitochondrial dysfunction, which led to the release of cytochrome c, caspase activation, and apoptosis (Mouria et al., 2002). Arem et al. studied 2,379 patients diagnosed with pancreatic cancer. This study failed to support the hypothesis of an association between the total intake of polyphenols or any type of flavonoid and the risk of pancreatic cancer (Arem et al., 2013). Furthermore, Molina-Molina-Montes et al. reported no association between the intake of flavonoids, flavonoid subclasses, or lignans and pancreatic cancer risk (Molina-Montes et al., 2016).
To the authors’ knowledge, no systematic review or meta-analysis has been conducted to assess the association between polyphenol intake and the risk of pancreatic cancer. Therefore, this study aimed to investigate the association between polyphenol intake and pancreatic cancer odds by conducting a systematic review and meta-analysis of observational studies.
Materials and Methods
Study protocol
This review was reported based on the Meta-Analysis of Observational Studies in Epidemiology (MOOSE) (Stroup et al., 2000) and Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines (Moher et al., 2010). The protocol was registered in PROSPERO (International Prospective Register of Systematic Reviews, registration number: CRD42023487302).
Search strategy
Articles published until April 2023 in PubMed, Scopus, and the Web of Science (ISI) were searched. The inclusion and inclution criteria were shown in Table 1. There were no restrictions on the language or publication dates. Furthermore, to find any other related articles, a manual, comprehensive search in Google Scholar and searches of the references of related published articles were performed. The search was updated to August 2024 to include any relevant published studies.
Eligibility criteria
Two independent researchers (Forootani B. and Yekrang Safakar H) screened the relevant studies according to the eligibility criteria, and contradictions were resolved by a third investigator (Salehi-Abargouei A). Studies were included in this review if they 1) were a prospective cohort (cohort, case-cohort, or nested case-control), case-control, or cross-sectional; 2) investigated the association between polyphenol intake as exposure and pancreatic cancer odds as an outcome; 3) were performed in adults; and 4) had enough information about the desired outcomes (odds ratio (OR), relative risk (RR), hazard ratio (HR), and 95% confidence interval (CI)) for polyphenols of interest. Moreover, animal studies, randomized control trials, and studies without the necessary data for extraction were excluded.
| Table 1. PICOS criteria for inclusion and exclusion of studies. | ||
| Criteria | Inclusion | Exclusion |
| Population | Adults (18 or more), regardless of health status | Animal studies, randomized control trials, and studies that did not have the necessary data for extraction were excluded |
| Intervention (Exposure) | Higher intake of polyphenols | |
| Comparator | Lower intake of polyphenols | |
| Outcome | Pancreatic cancer | |
| Study design | Observational studies | |
Data extraction
The necessary data for eligible studies, including the author’s last name, publication year, study name, location, sample size, design, type of polyphenol, age, sex, association measures, and confounding variables, were extracted independently by two investigators (Goodarzi S and Mirjalili F). The OR, RR, HR, and 95% CI were extracted for different quantile of polyphenol intake. The highest and lowest quantile of polyphenol intake were considered.
Quality assessment and grading of recommendations
Two investigators (Goodarzi S and Mirjalili F) assessed the quality of the included articles separately using the modified Newcastle-Ottawa scale (NOS) for cohort and case-control studies. This tool has several domains for evaluating the risk of bias, including selection, comparability, and outcome for cohort studies, and selection, comparability, and exposure for case-control studies. Each domain was assigned a maximum of four, two, and three points. According to the NOS( scores of 0–3 indicate poor quality, scores of 4–6 indicate fair quality, and scores of 7–9 indicate high quality (Stang, 2010).
The GRADE assessment for developing recommendations was performed using GRADE Pro software (https://gdt.gradepro.org/app/) (Guyatt et al., 2008). The GRADE assessment and recommendation were measured by Goodarzi S debated and revised by a second investigator (Salehi-Abargouei A), and discussed and agreed upon by all authors.
Statistical methods
The authors assessed the association between high polyphenol intake and the risk of pancreatic cancer. All data were converted to log ORs with standard errors (SEs) using the OR, RR, HR, and 95% CI as effect sizes for the meta-analyses. A random-effects model was used to pool effect sizes. The heterogeneity between studies was determined using the Q Cochrane test and I2 statistic (Higgins and Thompson, 2002). An I value ≥ 50% was considered to indicate heterogeneity and was reduced using a random-effects model. To estimate the impact of each study on the overall effect size, a sensitivity analysis was performed using the leave-one-out method as follows: one study was eliminated each time, and the analysis was performed again. To distinguish publication bias from other sample-size-related effects, funnel plots, Begg's rank correlation, and Egger's weighted regression tests were used (Deeks et al., 2005). If evidence of publication bias was observed, Duval & Tweedie's “trim and fill” analysis was performed to adjust for the effects of publication bias (Duval and Tweedie, 2000). This meta-analysis was performed using STATA software, version 17.0. The significance of the probability value (p-value) was also set at < 0.05.
Results
A total of 14230 reports were identified, 3790 duplicate records were removed, 9521 records were marked as ineligible after title and abstract screening, and 651 studies were excluded because they were irrelevant to this meta-analysis or did not meet the eligibility criteria. After reviewing the full texts of the articles, 260 studies were removed for the following reasons: 259 reports of other cancers and one study did not have enough data. Finally, 8 articles that met the eligibility criteria were included in the evaluation and analysis. A complete explanation of the study selection process is shown in Figure 1.
Characteristics of the included studies
Eligible studies are presented in Table 2. The total number of participants involved in the analysis was 1350913, with 5212 cases of pancreatic cancer. The age range of the population was 25-80. These studies were published between 2002 and 2020 and were performed in Italy (Rossi et al., 2012), the United States (4 studies) (Arem et al., 2013, Arts et al., 2002, Cutler et al., 2008, Nöthlings et al., 2007), Finland (Bobe et al., 2008), European countries (Molina-Montes et al., 2016) and Japan (Yamagiwa et al., 2020). One study was conducted exclusively on male participants (Bobe et al., 2008), two on female participants (Arts et al., 2002, Cutler et al., 2008) and the rest of the studies included both sexes. One study had a case-control design, whereas seven studies had cohort designs. The follow-up duration ranged from 8 (Nöthlings et al., 2007) to 18 (Cutler et al., 2008) years. All investigations used a validated food frequency questionnaire to assess dietary polyphenol intake, and the highest and lowest intakes of polyphenols were considered.
The necessary data for eligible studies, including the author’s last name, publication year, study name, location, sample size, design, type of polyphenol, age, sex, association measures, and confounding variables, were extracted independently by two investigators (Goodarzi S and Mirjalili F). The OR, RR, HR, and 95% CI were extracted for different quantile of polyphenol intake. The highest and lowest quantile of polyphenol intake were considered.
Quality assessment and grading of recommendations
Two investigators (Goodarzi S and Mirjalili F) assessed the quality of the included articles separately using the modified Newcastle-Ottawa scale (NOS) for cohort and case-control studies. This tool has several domains for evaluating the risk of bias, including selection, comparability, and outcome for cohort studies, and selection, comparability, and exposure for case-control studies. Each domain was assigned a maximum of four, two, and three points. According to the NOS( scores of 0–3 indicate poor quality, scores of 4–6 indicate fair quality, and scores of 7–9 indicate high quality (Stang, 2010).
The GRADE assessment for developing recommendations was performed using GRADE Pro software (https://gdt.gradepro.org/app/) (Guyatt et al., 2008). The GRADE assessment and recommendation were measured by Goodarzi S debated and revised by a second investigator (Salehi-Abargouei A), and discussed and agreed upon by all authors.
Statistical methods
The authors assessed the association between high polyphenol intake and the risk of pancreatic cancer. All data were converted to log ORs with standard errors (SEs) using the OR, RR, HR, and 95% CI as effect sizes for the meta-analyses. A random-effects model was used to pool effect sizes. The heterogeneity between studies was determined using the Q Cochrane test and I2 statistic (Higgins and Thompson, 2002). An I value ≥ 50% was considered to indicate heterogeneity and was reduced using a random-effects model. To estimate the impact of each study on the overall effect size, a sensitivity analysis was performed using the leave-one-out method as follows: one study was eliminated each time, and the analysis was performed again. To distinguish publication bias from other sample-size-related effects, funnel plots, Begg's rank correlation, and Egger's weighted regression tests were used (Deeks et al., 2005). If evidence of publication bias was observed, Duval & Tweedie's “trim and fill” analysis was performed to adjust for the effects of publication bias (Duval and Tweedie, 2000). This meta-analysis was performed using STATA software, version 17.0. The significance of the probability value (p-value) was also set at < 0.05.
Results
A total of 14230 reports were identified, 3790 duplicate records were removed, 9521 records were marked as ineligible after title and abstract screening, and 651 studies were excluded because they were irrelevant to this meta-analysis or did not meet the eligibility criteria. After reviewing the full texts of the articles, 260 studies were removed for the following reasons: 259 reports of other cancers and one study did not have enough data. Finally, 8 articles that met the eligibility criteria were included in the evaluation and analysis. A complete explanation of the study selection process is shown in Figure 1.
Characteristics of the included studies
Eligible studies are presented in Table 2. The total number of participants involved in the analysis was 1350913, with 5212 cases of pancreatic cancer. The age range of the population was 25-80. These studies were published between 2002 and 2020 and were performed in Italy (Rossi et al., 2012), the United States (4 studies) (Arem et al., 2013, Arts et al., 2002, Cutler et al., 2008, Nöthlings et al., 2007), Finland (Bobe et al., 2008), European countries (Molina-Montes et al., 2016) and Japan (Yamagiwa et al., 2020). One study was conducted exclusively on male participants (Bobe et al., 2008), two on female participants (Arts et al., 2002, Cutler et al., 2008) and the rest of the studies included both sexes. One study had a case-control design, whereas seven studies had cohort designs. The follow-up duration ranged from 8 (Nöthlings et al., 2007) to 18 (Cutler et al., 2008) years. All investigations used a validated food frequency questionnaire to assess dietary polyphenol intake, and the highest and lowest intakes of polyphenols were considered.
.PNG)
| Table 2. Epidemiologic studies on polyphenols intake in association with pancreatic cancer risk. | |||||||||
| First author, (Year), Country |
Design/Follow- Up (year) |
Cases/Controls or Cohort size |
Sex |
Age, dietary assessment |
Polyphenols |
Contrast |
Adjusted OR |
Adjustments |
Quality Score |
| Rossi et al. (2012), Italy. |
Case‒control/- |
326/652 |
F, M |
80.34, FFQ |
Flavanols, Flavanones, Flavonols, Flavones Anthocyanidins, Proanthocyanidins |
Q5 vs Q1 |
0.63(0.38-1.03) 0.68(0.41-1.14) 0.69(0.42-1.13) 0.88(0.53-1.46) 0.83(0.43-1.60) |
Age, Gender, Center of study, Year of the interview, Education, History of diabetes, Tobacco smoking, Alcohol drinking, and Nonalcohol energy intake. |
6 |
| Arem et al. (2013), USA. |
Prospective-Cohort/10.6 |
2379/537104 |
F, M |
71.50, FFQ |
Total flavonoids, Flavan-3-ols, Flavones, Flavonols Flavanones, Anthocyanidins, Isoflavones |
Q5 vs Q1 |
1.09(0.96-1.24) 1.03(0.91-1.17) 1.09(0.95-1.25) 1.09(0.95-1.24) 1.06(0.92-1.21) 1.10(0.96-1.27) 0.96(0.84-1.09) |
Age, Gender, Smoking, Diabetes, BMI, Alcohol, Calories, Saturated fat and Red meat intake |
8 |
| Bobe et al. (2008), Finland. |
Prospective-Cohort/16.1 |
306/27111 |
M |
69.50, FFQ |
Total flavonoids, Flavan-3-ols, Flavones, Flavonols Kaempferol, Myricetin Quercetin, Catechin Epicatechin, Apigenin, Luteolin |
Q5 vs Q1 |
0.90(0.64-1.28) 0.92(0.64-1.31) 0.99(0.70-1.42) 0.91(0.64-1.30) 0.93(0.65-1.33) 1.04(0.73-1.49) 1.07(0.75-1.53) 0.95(0.66-1.36) 0.90(0.63-1.28) 1.06(0.74-1.51) 1.09(0.77-1.56) |
Age, Years of smoking, Total number of cigarettes per day, Self-reported history of diabetes mellitus, and Energy-adjusted saturated fat intake. |
7 |
| Molina-Montes et al. (2016), EU countries. |
Prospective-Cohort/11.3 |
865/477309 |
F, M |
70.25, FFQ |
Total flavonoids, Flavan-3-ols, Flavones, Flavonols Flavanones, Anthocyanidins, Flavanols Proanthocyanidins, Anthocyanidins, Isoflavones Lignans, Theaflavins |
Q5 vs Q1 Q4 vs Q1 |
1.10(0.85-1.42) 1.23(0.93-1.62) 0.95(0.73-1.25) 1.31(1.00-1.72) 0.84(0.66-1.07) 1.02(0.78-1.33) 1.13(0.87-1.46) 1.02(0.80-1.31) 1.02(0.78-1.33) 0.91(0.64-1.28) 0.99(0.74-1.34) 1.35(1.03-1.75) |
Age, Gender, Centre, Total energy intake from fat and nonfat sources, BMI, Smoking status and intensity, Alcohol intake, Diabetes status at recruitment |
5 |
| Nothlings et al. (2007), USA. |
Prospective- Cohort/8 |
529/183518 |
F, M |
75.45, FFQ |
Flavonols, Quercetin Kaempferol, Myricetin Flavonols, Quercetin Kaempferol, Myricetin Flavonols, Quercetin Kaempferol, Myricetin |
Q5 vs Q1 Q4 vs Q1 Q4 vs Q1 |
0.77(0.58-1.03) 0.80(0.60-1.06) 0.78(0.58-1.05) 0.85(0.65-1.12) 0.78(0.55-1.13) 0.79(0.55-1.14) 0.72(0.49-1.04) 0.72(0.50-1.03) 0.87(0.60-1.25) 0.85(0.58-1.23) 0.87(0.60-1.27) 0.97(0.69-1.37) |
Age, History of diabetes mellitus, Family history of pancreatic cancer, BMI, Smoking status, Processed and red meat intake, Energy intake |
6 |
| Arts et al. (2002), USA. |
Prospective-Cohort/13 |
130/33339 |
F |
69.55, FFQ |
Catechins |
Q4 vs Q1 |
0.74(0.46-1.20) |
Age, Total energy intake, BMI, Waist-to-hip ratio, Physical activity, Smoking, Alcoho, fruit and vegetable. | 5 |
| Yamagiwa et al. (2020), Japan. |
Prospective- Cohort/16.9 |
577/90185 263/48286 314/41899 |
F, M |
69.40, FFQ |
Genistein, Genistein, Genistein |
Q4 vs Q1 |
1.33(1.03-1.73) 1.46(1.00-2.12) 1.22(0.85-1.74) |
Age, Gender, Smoking, Physical activity, History of diabetes mellitus, Family history of pancreatic cancer, BMI, intak of Ethanol, Fish, Meat, Vegetable, Fruit, Coffee, and Energy. | 8 |
| Cutler et al. (2008), USA. |
Prospective-Cohort/18 |
230/34708 |
F |
69.55, FFQ |
Total flavonoid, Isoflavones Anthocyanidins, Flavones Flavanones, Flavonols Flavan-3-ols, Proanthocyanidins, Total, Proanthocyanidins |
NR |
NR |
Age, Gender, Daily energy intake, Education level, Race, BMI, Multivitamin use, Activity level, Smoking history, and Pack years. |
6 |
| M: Male; F: Female; FFQ: Food frequency questionnaire; Q: Quantile; BMI: Body mass index; NR: Not reported; USA: United States of America | |||||||||
Quality assessment
Studies were identified as poor, fair, or high quality according to the modified NOS thresholds, of 0-3, 4-6, and 7-9, respectively (Stang, 2010). The authors classified three studies as high (Arem et al., 2013, Bobe et al., 2008, Yamagiwa et al., 2020) and five as fair (Arts et al., 2002, Cutler et al., 2008, Molina-Montes et al., 2016, Nöthlings et al., 2007, Rossi et al., 2012).
Meta-analysis
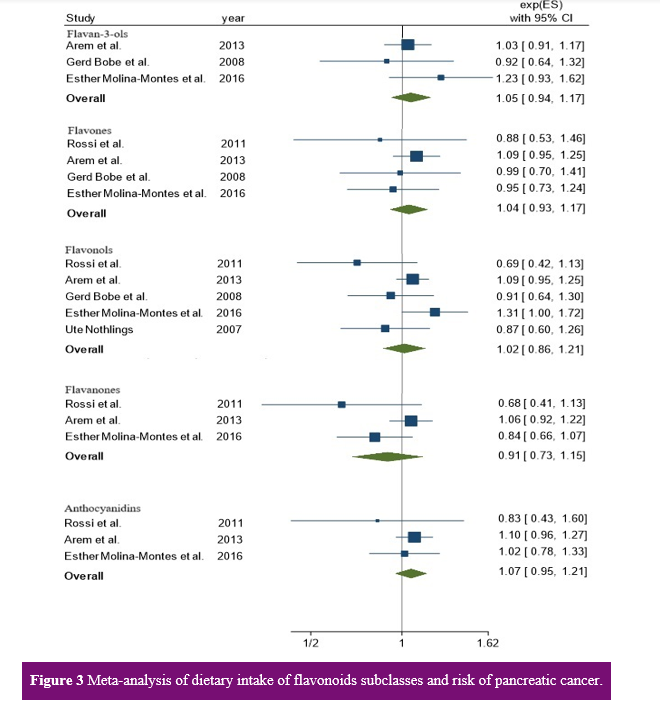
Dietary polyphenol intake and pancreatic cancer odds: Three articles with cohort designs were included in the analysis of total flavonoid intake and flavan-3-ols to the odds of pancreatic cancer (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016). The mean follow-up time for the pooled studies was 12.6 years (range 10.6-16.1 years). No associations between total flavonoid (Q5 vs. Q1: OR=1.07, 95% CI: 0.96, 1.19, I2=0.00%, P-value for heterogeneity=0.58) or flavan-3-ol (Q5 vs. Q1: OR=1.05, 95% CI: 0.94, 1.17, I2=0.00%, P-value for heterogeneity=0.39) intake and cancer risk were detected, with no evidence of heterogeneity (Figures 2 and 3).
Four studies (three cohorts and one case-control) were included in the analysis of the association between flavones and the odds of pancreatic cancer (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Rossi et al., 2012). The mean follow-up time was 12.6 years (range 10.6-16.1 years). Furthermore, there was no significant difference in the odds of pancreatic cancer according to the highest intake of flavones compared to the lowest intake (Q5 vs. Q1: OR=1.04, 95% CI: 0.93, 1.17; Figure 3), with no evidence of heterogeneity (I2=0.00%, P=0.71).
Five studies (four cohorts and one case-control) assessed the association between flavonol intake and pancreatic cancer risk (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Nöthlings et al., 2007, Rossi et al., 2012). The mean follow-up time was 11.5 years (range 8-16.1 years). The pooled OR for pancreatic cancer, comparing the highest versus lowest flavonol intake quintiles, was also Q5 vs. Q1 (OR=1.02, 95% CI: 0.86, 1.21; Figure 3), and the heterogeneity was low (I2=44.66%, P=0.12).
Three articles (two cohort and one case-control) were included in the analysis of flavanone and anthocyanidin intake concerning the pancreatic cancer odds (Arem et al., 2013, Molina-Montes et al., 2016, Rossi et al., 2012). The mean follow-up time for these studies was 11 years (range 10.6-11.3 years). The overall ORs for flavanone and anthocyanidin intake to the risk of pancreatic cancer were as follows: Q5 vs. Q1: OR=0.91, 95% CI: 0.73, 1.15, with high heterogeneity (I2=58.21%, P=0.09); and Q5 vs. Q1: OR=1.07, 95% CI: 0.95, 1.21, Figure 3, with low heterogeneity (I2=0.00%, P=0.65).
Qualitative analysis
In a study by Cutler et al., there was no association between pancreatic cancer incidence and total flavonoids, flavanones, anthocyanidins, flavones, flavonols, and flavan-3-ols in postmenopausal females (Cutler et al., 2008). Arem et al., Molina-Montes et al., and Cutler et al. reported that isoflavones are not related to the incidence of pancreatic cancer (Arem et al., 2013, Cutler et al., 2008, Molina-Montes et al., 2016). In one study, an inverse association was observed between the odds of pancreatic cancer and the use of proanthocyanidins for proanthocyanidins more than 3-mers (Rossi et al., 2012). Moreover, two studies did not demonstrate this relationship (Cutler et al., 2008, Molina-Montes et al., 2016). There was no relationship between kaempferol, myricetin, or quercetin and pancreatic cancer according to Gerd Bobe et al. and Ute Nothlings et al (Bobe et al., 2008, Nöthlings et al., 2007). Additionally, two studies did not observe this relationship with catechins (Arts et al., 2002, Bobe et al., 2008), and no association between flavanols and pancreatic cancer was observed in these two studies (Molina-Montes et al., 2016, Rossi et al., 2012). The remaining polyphenols, including epicatechin, apigenin, luteolin, and lignans, did not show any association (Bobe et al., 2008, Molina-Montes et al., 2016).
Studies were identified as poor, fair, or high quality according to the modified NOS thresholds, of 0-3, 4-6, and 7-9, respectively (Stang, 2010). The authors classified three studies as high (Arem et al., 2013, Bobe et al., 2008, Yamagiwa et al., 2020) and five as fair (Arts et al., 2002, Cutler et al., 2008, Molina-Montes et al., 2016, Nöthlings et al., 2007, Rossi et al., 2012).
Meta-analysis
Dietary polyphenol intake and pancreatic cancer odds: Three articles with cohort designs were included in the analysis of total flavonoid intake and flavan-3-ols to the odds of pancreatic cancer (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016). The mean follow-up time for the pooled studies was 12.6 years (range 10.6-16.1 years). No associations between total flavonoid (Q5 vs. Q1: OR=1.07, 95% CI: 0.96, 1.19, I2=0.00%, P-value for heterogeneity=0.58) or flavan-3-ol (Q5 vs. Q1: OR=1.05, 95% CI: 0.94, 1.17, I2=0.00%, P-value for heterogeneity=0.39) intake and cancer risk were detected, with no evidence of heterogeneity (Figures 2 and 3).
Four studies (three cohorts and one case-control) were included in the analysis of the association between flavones and the odds of pancreatic cancer (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Rossi et al., 2012). The mean follow-up time was 12.6 years (range 10.6-16.1 years). Furthermore, there was no significant difference in the odds of pancreatic cancer according to the highest intake of flavones compared to the lowest intake (Q5 vs. Q1: OR=1.04, 95% CI: 0.93, 1.17; Figure 3), with no evidence of heterogeneity (I2=0.00%, P=0.71).
Five studies (four cohorts and one case-control) assessed the association between flavonol intake and pancreatic cancer risk (Arem et al., 2013, Bobe et al., 2008, Molina-Montes et al., 2016, Nöthlings et al., 2007, Rossi et al., 2012). The mean follow-up time was 11.5 years (range 8-16.1 years). The pooled OR for pancreatic cancer, comparing the highest versus lowest flavonol intake quintiles, was also Q5 vs. Q1 (OR=1.02, 95% CI: 0.86, 1.21; Figure 3), and the heterogeneity was low (I2=44.66%, P=0.12).
Three articles (two cohort and one case-control) were included in the analysis of flavanone and anthocyanidin intake concerning the pancreatic cancer odds (Arem et al., 2013, Molina-Montes et al., 2016, Rossi et al., 2012). The mean follow-up time for these studies was 11 years (range 10.6-11.3 years). The overall ORs for flavanone and anthocyanidin intake to the risk of pancreatic cancer were as follows: Q5 vs. Q1: OR=0.91, 95% CI: 0.73, 1.15, with high heterogeneity (I2=58.21%, P=0.09); and Q5 vs. Q1: OR=1.07, 95% CI: 0.95, 1.21, Figure 3, with low heterogeneity (I2=0.00%, P=0.65).
Qualitative analysis
In a study by Cutler et al., there was no association between pancreatic cancer incidence and total flavonoids, flavanones, anthocyanidins, flavones, flavonols, and flavan-3-ols in postmenopausal females (Cutler et al., 2008). Arem et al., Molina-Montes et al., and Cutler et al. reported that isoflavones are not related to the incidence of pancreatic cancer (Arem et al., 2013, Cutler et al., 2008, Molina-Montes et al., 2016). In one study, an inverse association was observed between the odds of pancreatic cancer and the use of proanthocyanidins for proanthocyanidins more than 3-mers (Rossi et al., 2012). Moreover, two studies did not demonstrate this relationship (Cutler et al., 2008, Molina-Montes et al., 2016). There was no relationship between kaempferol, myricetin, or quercetin and pancreatic cancer according to Gerd Bobe et al. and Ute Nothlings et al (Bobe et al., 2008, Nöthlings et al., 2007). Additionally, two studies did not observe this relationship with catechins (Arts et al., 2002, Bobe et al., 2008), and no association between flavanols and pancreatic cancer was observed in these two studies (Molina-Montes et al., 2016, Rossi et al., 2012). The remaining polyphenols, including epicatechin, apigenin, luteolin, and lignans, did not show any association (Bobe et al., 2008, Molina-Montes et al., 2016).
.PNG)

Sensitivity analysis
According to the sensitivity analysis, by removing one study each time, the results did not change significantly for polyphenols, which showed that the results for total flavonoids, flavan-3-ols, flavones, flavonols, flavanones, and anthocyanidins were consistent and reliable.
Publication bias
An overview of the Begg funnel plot did not show any asymmetry in the meta-analysis of the total flavonoid, flavan-3-ol, flavone, or anthocyanidin intake to the odds of pancreatic cancer. No evidence of substantial publication bias was identified for total flavonoids (P=0.45 Begg’s test; P=0.29 for Egger’s test), flavan-3-ols (P=0.96, Begg’s test; P=1.00, Egger’s test), flavones (P=0.27, Begg’s test; P=0.73, Egger’s test), or anthocyanins (P=0.35, Begg’s test; P=0.29, Egger’s test). Although Begg’s test for flavonol intake and cancer risk was significant (P=0.08), Egger’s test did not confirm bias (P=0.13). Egger’s test for the association between flavones and cancer revealed a significant publication bias (P=0.03), but Begg’s test was not significant (P=0.29). Using the "trim and fill" method, two potentially missing studies were imputed for flavonol and flavanone intake, and no significant change in overall associations was observed.
In summary, the overall association between flavonoids and pancreatic cancer risk using
the random effects model is shown in
Table 3.
According to the sensitivity analysis, by removing one study each time, the results did not change significantly for polyphenols, which showed that the results for total flavonoids, flavan-3-ols, flavones, flavonols, flavanones, and anthocyanidins were consistent and reliable.
Publication bias
An overview of the Begg funnel plot did not show any asymmetry in the meta-analysis of the total flavonoid, flavan-3-ol, flavone, or anthocyanidin intake to the odds of pancreatic cancer. No evidence of substantial publication bias was identified for total flavonoids (P=0.45 Begg’s test; P=0.29 for Egger’s test), flavan-3-ols (P=0.96, Begg’s test; P=1.00, Egger’s test), flavones (P=0.27, Begg’s test; P=0.73, Egger’s test), or anthocyanins (P=0.35, Begg’s test; P=0.29, Egger’s test). Although Begg’s test for flavonol intake and cancer risk was significant (P=0.08), Egger’s test did not confirm bias (P=0.13). Egger’s test for the association between flavones and cancer revealed a significant publication bias (P=0.03), but Begg’s test was not significant (P=0.29). Using the "trim and fill" method, two potentially missing studies were imputed for flavonol and flavanone intake, and no significant change in overall associations was observed.
In summary, the overall association between flavonoids and pancreatic cancer risk using
the random effects model is shown in
Table 3.
| Table 3. Meta-analysis showing the overall association between flavonoids and pancreatic cancer risk, using random effects model. |
|||||||
| Exposure | Number of Studies |
Meta-analysis | Heterogeneity | Publication bias | |||
| Effect size (95% CI) |
P-value | I2 (%) |
|||||
| Effect | within heterogeneity |
Begg’s test | Egger’s test | ||||
| Total flavonoid | 3 | 1.07(0.96,1.19) | 0.21 | 0.58 | 0.29 | 0.45 | 0.00 |
| Flavan-3-ols | 3 | 1.05(0.94,1.17) | 0.40 | 0.39 | 1.00 | 0.96 | 0.00 |
| Flavones | 4 | 1.04(0.93,1.17) | 0.47 | 0.71 | 0.73 | 0.27 | 0.00 |
| Flavonols | 5 | 1.02(0.86,1.21) | 0.83 | 0.12 | 0.08 | 0.13 | 44.66 |
| Flavanones | 3 | 0.91(0.73,1.15) | 0.43 | 0.09 | 0.29 | 0.03 | 58.21 |
| Anthocyanidins | 3 | 1.07(0.95,1.21) | 0.26 | 0.65 | 0.29 | 0.35 | 0.00 |
| P-value effect < 0.05 is significant, P-value Egger’s test and Begg’s test < 0.1 is significant. |
|||||||
Discussion
This study investigated the association between polyphenol intake and pancreatic cancer odds in eight observational studies with 1384252 participants and 5342 pancreatic cancer. This systematic review and meta-analysis revealed no relationship between polyphenol intake and pancreatic cancer odds; however, in two studies, a positive relationship was observed between genistein and theaflavin intake and pancreatic cancer risk.
The authors are unaware of any systematic reviews or meta-analyses published in this field. In line with the results, Arem et al., Molina-Montes et al., and Cutler et al. reported no association between total flavonoid intake or any flavonoid subtypes and pancreatic cancer risk (Arem et al., 2013, Cutler et al., 2008, Molina-Montes et al., 2016). Although Rossi et al. demonstrated in a case-control study that dietary proanthocyanidins may confer some protection against pancreatic cancer risk, another subclass of flavonoids did not confirm this effect (Rossi et al., 2012). The association between catechin intake and pancreatic cancer incidence reported by Arts et al. was not statistically significant (Arts et al., 2002). A pooled analysis of 15 prospective cohorts, including 52,680 participants and 3,205 prostate cancer patients, revealed no association between the consumption of fruits and vegetables as the main source of polyphenols and prostate cancer risk (Petimar et al., 2017).
This study investigated the association between polyphenol intake and pancreatic cancer odds in eight observational studies with 1384252 participants and 5342 pancreatic cancer. This systematic review and meta-analysis revealed no relationship between polyphenol intake and pancreatic cancer odds; however, in two studies, a positive relationship was observed between genistein and theaflavin intake and pancreatic cancer risk.
The authors are unaware of any systematic reviews or meta-analyses published in this field. In line with the results, Arem et al., Molina-Montes et al., and Cutler et al. reported no association between total flavonoid intake or any flavonoid subtypes and pancreatic cancer risk (Arem et al., 2013, Cutler et al., 2008, Molina-Montes et al., 2016). Although Rossi et al. demonstrated in a case-control study that dietary proanthocyanidins may confer some protection against pancreatic cancer risk, another subclass of flavonoids did not confirm this effect (Rossi et al., 2012). The association between catechin intake and pancreatic cancer incidence reported by Arts et al. was not statistically significant (Arts et al., 2002). A pooled analysis of 15 prospective cohorts, including 52,680 participants and 3,205 prostate cancer patients, revealed no association between the consumption of fruits and vegetables as the main source of polyphenols and prostate cancer risk (Petimar et al., 2017).
Consistent with this study, Bobe et al. reported a significant relationship between total flavonoids, flavonols, flavan-3-ols, kaempferol, quercetin, catechin, and epicatechin and low pancreatic cancer risk and suggested that a flavonoid-rich diet may reduce the incidence of pancreatic cancer in smokers who do not consume supplemental A-tocopherol and/or B-carotene (Bobe et al., 2008). Similarly, Nothlings et al. demonstrated a protective effect of total flavonol intake on pancreatic cancer risk, particularly in current smokers (Nöthlings et al., 2007). Animal studies have shown that food-derived polyphenols, including quercetin and transresveratrol, inhibit pancreatic cancer development and prevent metastasis (Mouria et al., 2002). In both in vitro and in vivo models of pancreatic cancer stem cells, quercetin targets pancreatic cancer stem cells and inhibits their growth by reducing their proliferation, angiogenesis, cancer stem cell marker expression, and inducing apoptosis (Zhou et al., 2010). They reported that hispidulin, a small flavonoid molecule, suppresses angiogenesis and the development of human pancreatic cancer (He et al., 2011).
Studies have shown that polyphenol consumption increases the risk of pancreatic cancer. Yamagiwa reported that the highest intake of genistein was significantly positively associated with the incidence of pancreatic cancer (Yamagiwa et al., 2020).
Polyphenols have long been known as natural antioxidants and secondary plant metabolites, including over 4000 types that are grouped into nine categories: flavonoids, isoflavonoids, aurones, chalconoids, flavonolignans, lignans, stilbenoids, curcuminoids, and tannins. Flavonoids are divided into six subclasses: flavones, isoflavones, flavonols, flavanones, flavanols, and anthocyanins (Cory et al., 2018, Tešić et al., 2018). The anticancer mechanisms of these compounds include inhibition of proliferation, induction of apoptosis, and suppression of the cell cycle. Moreover, polyphenols can modulate signaling pathways and impact epigenetic modifications, such as DNA methylation and the expression patterns of microRNAs (miRNAs) (Nasir et al., 2022). Importantly, the bioavailability of polyphenols is low, and although polyphenol components exist in many foods, their health benefits hinge on the amount of ingested polyphenols, especially their bioavailability and bioaccessibility. The poor bioavailability of polyphenol components in fruit and vegetable matrices is due to their low bioaccessibility in the small intestine due to the physical and chemical interactions of polyphenols with the indigestible polysaccharides of cell walls (Bié et al., 2023) .
This study had several limitations. First, dietary assessment techniques and nutrient databases are influenced by measurement errors, which can lead to inaccurate risk assessment. Second, due to the small number of studies, subgroup analysis and meta-analysis of other polyphenol types, including quercetin, were not possible. Third, the adjustments for confounding variables were not the same across all studies. Fourth, the interaction between the highest intake of polyphenols in one study and the lowest intake in another may have affected the results. Notably, the quality of the meta-evidence in the current study was very low. Therefore, future investigations should be conducted to confirm these findings.
Conclusion
In conclusion, the authors did not observe a relationship between polyphenol intake and pancreatic cancer odds. However, polyphenols have poor bioavailability and bioaccessibility, which raises the question of whether the anticancer effect of polyphenols in vitro and in vivo is overstated or whether dietary intake of polyphenols does not have this effect and needs to be consumed as an extract. Notably, the type of polyphenol administered may also be important. Considering these findings, the results of this study should be interpreted with caution, and further high-quality research with a precise design needs to be performed to determine whether there is a relationship.
Authors’ contributions
Salehi-Abargoui A, Forootani B, and Yekrang Safakar H. contributed to the conception and design of this study. Material preparation, data collection, and analysis were performed by Goodarzi S, Mirjali F, Forootani B, and Yekrang Safakar H. The first draft of the manuscript was written by Goodarzi S and Mirjalili F, and all the authors commented on the previous versions of the manuscript. The final draft of the manuscript has been revised by Salehi-Abargouei A, who had primary responsibility for the final content. All the authors have read and approved the final manuscript.
Confilic of interests
The authors declared no conflict of interests .
Funding
This study was funded by the Research Center for Food Hygiene and Safety, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
References
Anderson LN, Cotterchio M & Gallinger S 2009. Lifestyle, dietary, and medical history factors associated with pancreatic cancer risk in Ontario, Canada. Cancer causes & control. 20 (6): 825-834.
Arem H, et al. 2013. Flavonoid intake and risk of pancreatic cancer in the National Institutes of Health-AARP Diet and Health Study Cohort. British journal of cancer. 108 (5): 1168-1172.
Arts I, Jacobs D, Gross M, Harnack L & Folsom A 2002. Dietary catechins and cancer incidence among postmenopausal women: the Iowa Women's Health Study (United States). Cancer causes & control : CCC. 13 (4): 373-382.
Bao N, Chen F & Dai D 2020. The regulation of host intestinal microbiota by polyphenols in the development and prevention of chronic kidney disease. Frontiers in immunology. 10: 2981.
Bié J, Sepodes B, Fernandes PC & Ribeiro MH 2023. Polyphenols in health and disease: Gut microbiota, bioaccessibility, and bioavailability. Compounds. 3 (1): 40-72.
Bobe G, et al. 2008. Flavonoid intake and risk of pancreatic cancer in male smokers (Finland). Faseb journal. 22.
Bray F, et al. 2018. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A cancer journal for clinicians. 68 (6): 394–424.
Chan JM, Wang F & Holly EA 2005. Vegetable and fruit intake and pancreatic cancer in a population-based case-control study in the San Francisco bay area. Cancer epidemiology biomarkers & prevention. 14 (9): 2093-2097.
Cory H, Passarelli S, Szeto J, Tamez M & Mattei J 2018. The role of polyphenols in human health and food systems: A mini-review. Frontiers in nutrition. 5: 87.
Cutler G, et al. 2008. Dietary flavonoid intake and risk of cancer in postmenopausal women: the Iowa Women's Health Study. International journal of cancer. 123 (3): 664-671.
David AVA, Arulmoli R & Parasuraman S 2016. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacognosy reviews. 10 (20): 84.
Deeks JJ, Macaskill P & Irwig L 2005. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. Journal of clinical epidemiology. 58 (9): 882-893.
Duval S & Tweedie R 2000. Trim and fill: a simple funnel‐plot–based method of testing and adjusting for publication bias in meta‐analysis. Biometrics. 56 (2): 455-463.
Ghadirian P & Nkondjock A 2010. Consumption of food groups and the risk of pancreatic cancer: a case–control study. Journal of gastrointestinal cancer. 41 (2): 121-129.
Goñi Cambrodón I & Hernández Galiot A 2019. Intake of nutrient and non-nutrient dietary antioxidants. contribution of macromolecular antioxidant polyphenols in an elderly mediterranean population. Nutrients. 11 (9): 2165.
Guyatt GH, et al. 2008. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. British medical journal. 336 (7650): 924-926.
He L, et al. 2011. Hispidulin, a small flavonoid molecule, suppresses the angiogenesis and growth of human pancreatic cancer by targeting vascular endothelial growth factor receptor 2‐mediated PI3K/Akt/mTOR signaling pathway. Cancer science. 102 (1): 219-225.
Higgins JP & Thompson SG 2002. Quantifying heterogeneity in a meta‐analysis. Statistics in medicine. 21 (11): 1539-1558.
Jansen RJ, et al. 2011. Fruit and vegetable consumption is inversely associated with having pancreatic cancer. Cancer causes & control. 22 (12): 1613-1625.
Klein AP 2021. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nature reviews gastroenterology & hepatology. 18 (7): 493-502.
Lichota A, Gwozdzinski L & Gwozdzinski K 2019. Therapeutic potential of natural compounds in inflammation and chronic venous insufficiency. European journal of medicinal chemistry. 176: 68-91.
Liu RH 2013. Health-promoting components of fruits and vegetables in the diet. Advances in nutrition. 4 (3): 384S-392S.
Liu S-Z, et al. 2014. Dietary factors and risk of pancreatic cancer: a multi-centre case-control study in China. Asian Pacific journal of cancer prevention. 15 (18): 7947-7950.
Maisonneuve P & Lowenfels AB 2010. Epidemiology of pancreatic cancer: an update. Digestive diseases. 28 (4-5): 645-656.
Maisonneuve P & Lowenfels AB 2015. Risk factors for pancreatic cancer: a summary review of meta-analytical studies. International journal of epidemiology. 44 (1): 186-198.
Michaud DS, et al. 2005. Dietary patterns and pancreatic cancer risk in men and women. Journal of the national cancer institute. 97 (7): 518-524.
Midha S, Chawla S & Garg PK 2016. Modifiable and non-modifiable risk factors for pancreatic cancer: A review. Cancer letters. 381 (1): 269-277.
Miean KH & Mohamed S 2001. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. Journal of agricultural and food chemistry. 49 (6): 3106-3112.
Moher D, Liberati A, Tetzlaff J, Altman DG & Group P 2010. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. International journal of surgery. 8 (5): 336-341.
Molina-Montes E, et al. 2016. Flavonoid and lignan intake and pancreatic cancer risk in the European prospective investigation into cancer and nutrition cohort. International journal of cancer. 139 (7): 1480-1492.
Mouria M, et al. 2002. Food‐derived polyphenols inhibit pancreatic cancer growth through mitochondrial cytochrome C release and apoptosis. International journal of cancer. 98 (5): 761-769.
Nasir A, et al. 2022. Nutrigenomics: Epigenetics and cancer prevention: A comprehensive review. Critical reviews in food science and nutrition. 60 (8): 1375-1387.
Nöthlings U, Murphy SP, Wilkens LR, Henderson BE & Kolonel LN 2007. Flavonols and pancreatic cancer risk: the multiethnic cohort study. American journal of epidemiology. 166 (8): 924-931.
Petimar J, et al. 2017. A pooled analysis of 15 prospective cohort studies on the association between fruit, vegetable, and mature bean consumption and risk of prostate cancer. Cancer epidemiology, biomarkers & prevention. 26 (8): 1276-1287.
Pourshams A, et al. 2019. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. lancet gastroenterology & hepatology. 4 (12): 934-947.
Rawla P, Sunkara T & Gaduputi V 2019. Epidemiology of pancreatic cancer: global trends, etiology and risk factors. World journal of oncology. 10 (1): 10.
Rossi M, et al. 2012. Proanthocyanidins and other flavonoids in relation to pancreatic cancer: a case-control study in Italy. Annals of oncology. 23 (6): 1488-1493.
Sanches-Silva A, et al. 2020. Therapeutic potential of polyphenols in cardiovascular diseases: Regulation of mTOR signaling pathway. Pharmacological research. 152: 104626.
Serra D, Almeida LM & Dinis TC 2020. Polyphenols in the management of brain disorders: Modulation of the microbiota-gut-brain axis. Advances in food and nutrition research. 91: 1-27.
Singh SS, et al. 2020. NF-κB-mediated neuroinflammation in Parkinson’s disease and potential therapeutic effect of polyphenols. Neurotoxicity research. 37 (3): 491-507.
Slavin JL & Lloyd B 2012. Health benefits of fruits and vegetables. Advances in nutrition. 3 (4): 506-516.
Slimestad R, Fossen T & Brede C 2020. Flavonoids and other phenolics in herbs commonly used in Norwegian commercial kitchens. Food chemistry. 309: 125678.
Stang A 2010. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. European journal of epidemiology. 25: 603-605.
Stroup DF, et al. 2000. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 283 (15): 2008-2012.
Tešić ŽL, Gašić UM & Milojković-Opsenica DM 2018. Polyphenolic profile of the fruits grown in Serbia. In Advances in Plant Phenolics: From Chemistry to Human Health, pp. 47-66. ACS Publications.
Tian B & Liu J 2020. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. Journal of the science of food and agriculture. 100 (4): 1392-1404.
Vuong Q, et al. 2014. Fruit-derived phenolic compounds and pancreatic cancer: Perspectives from Australian native fruits. Journal of ethnopharmacology. 152 (2): 227-242.
Wu Q-J, et al. 2016. Consumption of fruit and vegetables reduces risk of pancreatic cancer: evidence from epidemiological studies. European journal of cancer prevention. 25 (3): 196-205.
Yamagiwa Y, et al. 2020. Soy food intake and pancreatic cancer risk: The Japan public health center-based prospective study. Cancer epidemiology biomarkers and prevention. 29 (6): 1214-1221.
Zhou W, et al. 2010. Dietary polyphenol quercetin targets pancreatic cancer stem cells. International journal of oncology. 37 (3): 551-561.
This study had several limitations. First, dietary assessment techniques and nutrient databases are influenced by measurement errors, which can lead to inaccurate risk assessment. Second, due to the small number of studies, subgroup analysis and meta-analysis of other polyphenol types, including quercetin, were not possible. Third, the adjustments for confounding variables were not the same across all studies. Fourth, the interaction between the highest intake of polyphenols in one study and the lowest intake in another may have affected the results. Notably, the quality of the meta-evidence in the current study was very low. Therefore, future investigations should be conducted to confirm these findings.
Conclusion
In conclusion, the authors did not observe a relationship between polyphenol intake and pancreatic cancer odds. However, polyphenols have poor bioavailability and bioaccessibility, which raises the question of whether the anticancer effect of polyphenols in vitro and in vivo is overstated or whether dietary intake of polyphenols does not have this effect and needs to be consumed as an extract. Notably, the type of polyphenol administered may also be important. Considering these findings, the results of this study should be interpreted with caution, and further high-quality research with a precise design needs to be performed to determine whether there is a relationship.
Authors’ contributions
Salehi-Abargoui A, Forootani B, and Yekrang Safakar H. contributed to the conception and design of this study. Material preparation, data collection, and analysis were performed by Goodarzi S, Mirjali F, Forootani B, and Yekrang Safakar H. The first draft of the manuscript was written by Goodarzi S and Mirjalili F, and all the authors commented on the previous versions of the manuscript. The final draft of the manuscript has been revised by Salehi-Abargouei A, who had primary responsibility for the final content. All the authors have read and approved the final manuscript.
Confilic of interests
The authors declared no conflict of interests .
Funding
This study was funded by the Research Center for Food Hygiene and Safety, Shahid Sadoughi University of Medical Sciences, Yazd, Iran.
References
Anderson LN, Cotterchio M & Gallinger S 2009. Lifestyle, dietary, and medical history factors associated with pancreatic cancer risk in Ontario, Canada. Cancer causes & control. 20 (6): 825-834.
Arem H, et al. 2013. Flavonoid intake and risk of pancreatic cancer in the National Institutes of Health-AARP Diet and Health Study Cohort. British journal of cancer. 108 (5): 1168-1172.
Arts I, Jacobs D, Gross M, Harnack L & Folsom A 2002. Dietary catechins and cancer incidence among postmenopausal women: the Iowa Women's Health Study (United States). Cancer causes & control : CCC. 13 (4): 373-382.
Bao N, Chen F & Dai D 2020. The regulation of host intestinal microbiota by polyphenols in the development and prevention of chronic kidney disease. Frontiers in immunology. 10: 2981.
Bié J, Sepodes B, Fernandes PC & Ribeiro MH 2023. Polyphenols in health and disease: Gut microbiota, bioaccessibility, and bioavailability. Compounds. 3 (1): 40-72.
Bobe G, et al. 2008. Flavonoid intake and risk of pancreatic cancer in male smokers (Finland). Faseb journal. 22.
Bray F, et al. 2018. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A cancer journal for clinicians. 68 (6): 394–424.
Chan JM, Wang F & Holly EA 2005. Vegetable and fruit intake and pancreatic cancer in a population-based case-control study in the San Francisco bay area. Cancer epidemiology biomarkers & prevention. 14 (9): 2093-2097.
Cory H, Passarelli S, Szeto J, Tamez M & Mattei J 2018. The role of polyphenols in human health and food systems: A mini-review. Frontiers in nutrition. 5: 87.
Cutler G, et al. 2008. Dietary flavonoid intake and risk of cancer in postmenopausal women: the Iowa Women's Health Study. International journal of cancer. 123 (3): 664-671.
David AVA, Arulmoli R & Parasuraman S 2016. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacognosy reviews. 10 (20): 84.
Deeks JJ, Macaskill P & Irwig L 2005. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. Journal of clinical epidemiology. 58 (9): 882-893.
Duval S & Tweedie R 2000. Trim and fill: a simple funnel‐plot–based method of testing and adjusting for publication bias in meta‐analysis. Biometrics. 56 (2): 455-463.
Ghadirian P & Nkondjock A 2010. Consumption of food groups and the risk of pancreatic cancer: a case–control study. Journal of gastrointestinal cancer. 41 (2): 121-129.
Goñi Cambrodón I & Hernández Galiot A 2019. Intake of nutrient and non-nutrient dietary antioxidants. contribution of macromolecular antioxidant polyphenols in an elderly mediterranean population. Nutrients. 11 (9): 2165.
Guyatt GH, et al. 2008. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. British medical journal. 336 (7650): 924-926.
He L, et al. 2011. Hispidulin, a small flavonoid molecule, suppresses the angiogenesis and growth of human pancreatic cancer by targeting vascular endothelial growth factor receptor 2‐mediated PI3K/Akt/mTOR signaling pathway. Cancer science. 102 (1): 219-225.
Higgins JP & Thompson SG 2002. Quantifying heterogeneity in a meta‐analysis. Statistics in medicine. 21 (11): 1539-1558.
Jansen RJ, et al. 2011. Fruit and vegetable consumption is inversely associated with having pancreatic cancer. Cancer causes & control. 22 (12): 1613-1625.
Klein AP 2021. Pancreatic cancer epidemiology: understanding the role of lifestyle and inherited risk factors. Nature reviews gastroenterology & hepatology. 18 (7): 493-502.
Lichota A, Gwozdzinski L & Gwozdzinski K 2019. Therapeutic potential of natural compounds in inflammation and chronic venous insufficiency. European journal of medicinal chemistry. 176: 68-91.
Liu RH 2013. Health-promoting components of fruits and vegetables in the diet. Advances in nutrition. 4 (3): 384S-392S.
Liu S-Z, et al. 2014. Dietary factors and risk of pancreatic cancer: a multi-centre case-control study in China. Asian Pacific journal of cancer prevention. 15 (18): 7947-7950.
Maisonneuve P & Lowenfels AB 2010. Epidemiology of pancreatic cancer: an update. Digestive diseases. 28 (4-5): 645-656.
Maisonneuve P & Lowenfels AB 2015. Risk factors for pancreatic cancer: a summary review of meta-analytical studies. International journal of epidemiology. 44 (1): 186-198.
Michaud DS, et al. 2005. Dietary patterns and pancreatic cancer risk in men and women. Journal of the national cancer institute. 97 (7): 518-524.
Midha S, Chawla S & Garg PK 2016. Modifiable and non-modifiable risk factors for pancreatic cancer: A review. Cancer letters. 381 (1): 269-277.
Miean KH & Mohamed S 2001. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. Journal of agricultural and food chemistry. 49 (6): 3106-3112.
Moher D, Liberati A, Tetzlaff J, Altman DG & Group P 2010. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. International journal of surgery. 8 (5): 336-341.
Molina-Montes E, et al. 2016. Flavonoid and lignan intake and pancreatic cancer risk in the European prospective investigation into cancer and nutrition cohort. International journal of cancer. 139 (7): 1480-1492.
Mouria M, et al. 2002. Food‐derived polyphenols inhibit pancreatic cancer growth through mitochondrial cytochrome C release and apoptosis. International journal of cancer. 98 (5): 761-769.
Nasir A, et al. 2022. Nutrigenomics: Epigenetics and cancer prevention: A comprehensive review. Critical reviews in food science and nutrition. 60 (8): 1375-1387.
Nöthlings U, Murphy SP, Wilkens LR, Henderson BE & Kolonel LN 2007. Flavonols and pancreatic cancer risk: the multiethnic cohort study. American journal of epidemiology. 166 (8): 924-931.
Petimar J, et al. 2017. A pooled analysis of 15 prospective cohort studies on the association between fruit, vegetable, and mature bean consumption and risk of prostate cancer. Cancer epidemiology, biomarkers & prevention. 26 (8): 1276-1287.
Pourshams A, et al. 2019. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. lancet gastroenterology & hepatology. 4 (12): 934-947.
Rawla P, Sunkara T & Gaduputi V 2019. Epidemiology of pancreatic cancer: global trends, etiology and risk factors. World journal of oncology. 10 (1): 10.
Rossi M, et al. 2012. Proanthocyanidins and other flavonoids in relation to pancreatic cancer: a case-control study in Italy. Annals of oncology. 23 (6): 1488-1493.
Sanches-Silva A, et al. 2020. Therapeutic potential of polyphenols in cardiovascular diseases: Regulation of mTOR signaling pathway. Pharmacological research. 152: 104626.
Serra D, Almeida LM & Dinis TC 2020. Polyphenols in the management of brain disorders: Modulation of the microbiota-gut-brain axis. Advances in food and nutrition research. 91: 1-27.
Singh SS, et al. 2020. NF-κB-mediated neuroinflammation in Parkinson’s disease and potential therapeutic effect of polyphenols. Neurotoxicity research. 37 (3): 491-507.
Slavin JL & Lloyd B 2012. Health benefits of fruits and vegetables. Advances in nutrition. 3 (4): 506-516.
Slimestad R, Fossen T & Brede C 2020. Flavonoids and other phenolics in herbs commonly used in Norwegian commercial kitchens. Food chemistry. 309: 125678.
Stang A 2010. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. European journal of epidemiology. 25: 603-605.
Stroup DF, et al. 2000. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 283 (15): 2008-2012.
Tešić ŽL, Gašić UM & Milojković-Opsenica DM 2018. Polyphenolic profile of the fruits grown in Serbia. In Advances in Plant Phenolics: From Chemistry to Human Health, pp. 47-66. ACS Publications.
Tian B & Liu J 2020. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. Journal of the science of food and agriculture. 100 (4): 1392-1404.
Vuong Q, et al. 2014. Fruit-derived phenolic compounds and pancreatic cancer: Perspectives from Australian native fruits. Journal of ethnopharmacology. 152 (2): 227-242.
Wu Q-J, et al. 2016. Consumption of fruit and vegetables reduces risk of pancreatic cancer: evidence from epidemiological studies. European journal of cancer prevention. 25 (3): 196-205.
Yamagiwa Y, et al. 2020. Soy food intake and pancreatic cancer risk: The Japan public health center-based prospective study. Cancer epidemiology biomarkers and prevention. 29 (6): 1214-1221.
Zhou W, et al. 2010. Dietary polyphenol quercetin targets pancreatic cancer stem cells. International journal of oncology. 37 (3): 551-561.
Type of article: review article |
Subject:
public specific
Received: 2025/03/10 | Published: 2026/05/30 | ePublished: 2026/05/30
Received: 2025/03/10 | Published: 2026/05/30 | ePublished: 2026/05/30
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |



